Unit 3 Lesson 10: BioFlexBook Questions
(1) What is a protein and what is its relationship to amino acids?
A protein is a molecule that living things use to provide different functions for the cell. The relationship is that each unique amino acid sequence makes a protein.
(2) Identify an analogy that fits the relationship that proteins have to monomers and polymers.
Monomers are molecules that can be bonded to other identical molecules to form a polymer. An analogy that could be used to fit that relationship is Legos because with one Lego you can't really make anything but if you add more and more Legos in a specific pattern you will end up with the product you want.
(3) What does denaturing mean and why does a denatured protein no longer function normally?
Denaturing is when a protein loses structure from an external stress. It can cause cell disfunction or cell death.
(4) What are the four structural levels of a protein?
The four structural levels of a protien are:
1. Primary Structure
2. Secondary Structure
3. Tertiary Structure
4. Quatenary Structure
(5) Why is the shape of a protein so critical to its proper functioning?
The shape is so crucial because the shape helps the protein do its job and when the shape is wrong it can not function properly.
A protein is a molecule that living things use to provide different functions for the cell. The relationship is that each unique amino acid sequence makes a protein.
(2) Identify an analogy that fits the relationship that proteins have to monomers and polymers.
Monomers are molecules that can be bonded to other identical molecules to form a polymer. An analogy that could be used to fit that relationship is Legos because with one Lego you can't really make anything but if you add more and more Legos in a specific pattern you will end up with the product you want.
(3) What does denaturing mean and why does a denatured protein no longer function normally?
Denaturing is when a protein loses structure from an external stress. It can cause cell disfunction or cell death.
(4) What are the four structural levels of a protein?
The four structural levels of a protien are:
1. Primary Structure
2. Secondary Structure
3. Tertiary Structure
4. Quatenary Structure
(5) Why is the shape of a protein so critical to its proper functioning?
The shape is so crucial because the shape helps the protein do its job and when the shape is wrong it can not function properly.
Lab Hypothesis
1. (Heat) The egg whites in heat will react first because heat denatures the protein shape the fastest.
2. (Salt) The egg whites will not denature because salt won't necessarily combine to become a compound and cause change.
3. (Baking soda) The egg whites in the sodium bicarbonate will denature the proteins because it will weaken the protein strands of the egg whites causing it to break down
4. (Lemon juice) The egg whites in the silver nitrate will denature the egg quickly because the silver will react with the egg whites and water, oxidizing the silver and changing its color.
5. (Alcohol) The egg whites in rubbing alcohol will denature the egg quickly because it takes a shorter amount of time for a liquid to combine with a liquid than a solid with a liquid.
6. (Silver nitrate) The egg whites in the lemon juice will not denature the eggs because the lemon and egg lipids will not combine to form a chemical reaction.
2. (Salt) The egg whites will not denature because salt won't necessarily combine to become a compound and cause change.
3. (Baking soda) The egg whites in the sodium bicarbonate will denature the proteins because it will weaken the protein strands of the egg whites causing it to break down
4. (Lemon juice) The egg whites in the silver nitrate will denature the egg quickly because the silver will react with the egg whites and water, oxidizing the silver and changing its color.
5. (Alcohol) The egg whites in rubbing alcohol will denature the egg quickly because it takes a shorter amount of time for a liquid to combine with a liquid than a solid with a liquid.
6. (Silver nitrate) The egg whites in the lemon juice will not denature the eggs because the lemon and egg lipids will not combine to form a chemical reaction.
Data/Lab Questions
Five Paragraph Essay on Lab
Nicolas Vasquez, Jake Ngu, Emilio Lopez, Robert Santos
Mr. Helms
Gen Bio 1D
11 December 2013
Denaturing Egg White
During this lab, we had the question of “What is the best way to denature proteins?” Proteins are essential to all organisms. They build both physical and chemical mass, help fight viruses and bacteria, and help replicate DNA. Denaturing is to take away or change one's natural properties and qualities. Denatured proteins no longer have correct structure. It no longer has its former functions or can perform them and is weakened. When proteins are denatured, they can no longer hold have their structure and no longer have their former functions. Denaturing is very common in one’s daily life. It occurs when we cook as well as in nature.
We believe that the proteins will denature the egg the fastest and most noticeably. Eggs are heated to destroy the bacteria to make it tastier and edible. We believed that heat is the best method for denaturing proteins in eggs, as this is the method most commonly used in the food industry.
In our lab, we started by separating four egg whites from their yolk, and putting the egg whites in a tub. We then separated the egg whites into six separate vials, which would be put under various levels of stress. The first vial was put into a pot of boiling water, salt was added to the second, baking soda to the third, lemon juice to the fourth, rubbing alcohol to the fifth, and silver nitrate to the sixth.
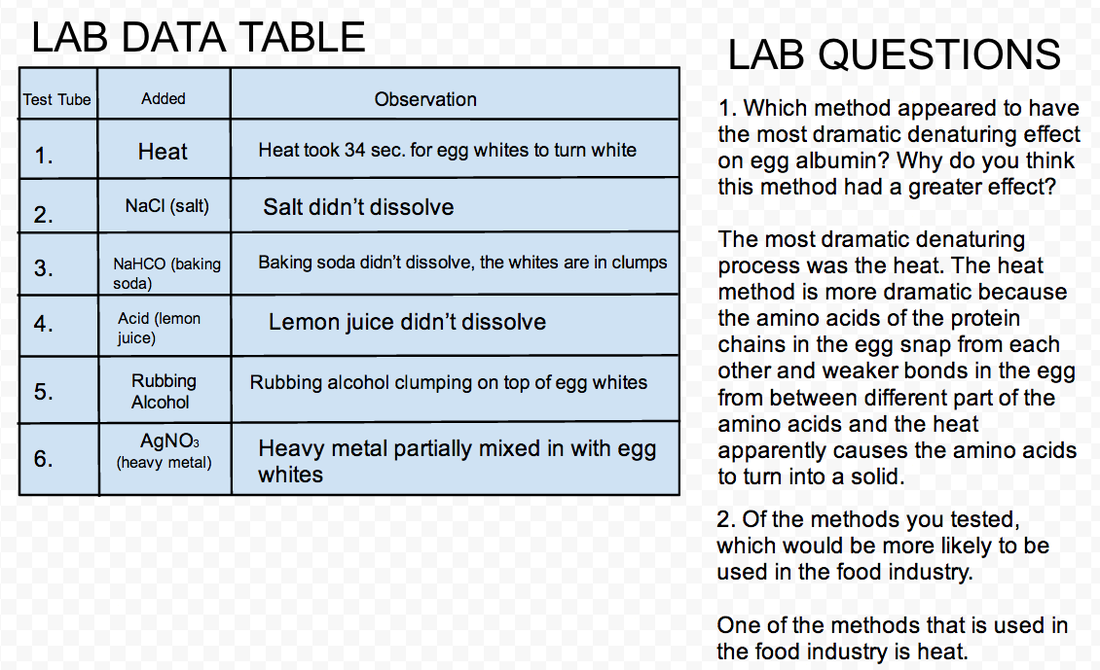
The reason for these certain results all have to do with the various denaturing methods. Heat causes the amino acids to break from the protein chain, and then the smaller weaker chains come and hold them together to the point where they become solid. The next thing is salt. Salt didn’t do anything to the eggs, but over time, salt tends to preserve things. This is because salt preserves the structures of the amino acids, specifically territory The baking soda also had no effect on the eggs. The lemon juice also had no effect. The rubbing alcohol and silver nitrate caused the egg whites to clump up on the top in wavy streaks.
The results in our lab during heat took 34 seconds to make the egg whites turn white. After the egg whites mixed in the NaCl(salt), the NaCl reacted by not dissolving in the whites. When we mixed the egg white with the NaHCO(Baking soda), the NaHCO inside the whites did not dissolve didn. After we mixed the lemon juice in with the egg whites, the lemon juice didn't react and neither did the egg white, the lemon juice simply just didn't dissolve in the egg white. When we added the rubbing alcohol into the egg white, all that happened was the rubbing alcohol clumped up on top of the egg whites. When we mixed the silver nitrate in with the egg whites, the white and the silver nitrate partially mixed together.